 Although an atom can gain electrons and become negatively charged or lose. The atomic number (number of protons) is a subscript at the lower left of the symbol of the element. The nuclear symbol indicates the composition of the nucleus. As per the IUPAC guidelines for the nomenclature of elements with atomic number greater than 100, the alternate name for Oganesson is. For instance, you might have read about it in discussions of nuclear energy, the Fukushima reactor tragedy, or the development of nuclear weapons. The periodic table of elements is widely used in the field of Chemistry to look up chemical elements as they are arranged in a manner that displays periodic trends in the chemical properties of the elements. Introduction Radioactivity pops up fairly often in the news. The atomic number indicates how many protons are in an element. The atomic number is the number of protons in an atom, and isotopes have the same atomic number but differ in the number of neutrons. Both the elements have different atomic number and different mass number but they have the same number of neutrons. The atomic number is different for every element present in a periodic table. The symbol for a specific isotope of any element is written by placing the mass number as a superscript to the left of the element symbol (see image below). Use a Periodic Table to look up the element with an atomic number of 32. * The given element is \(16 – 8 = 8 neutrons). The notation 3 d8 (read threedeight) indicates eight electrons in the d subshell (i.e., l 2) of the principal shell for which n 3. Exercise: Write the Nuclear Notation for the three stable isotopes of Neon which have 10. Sometimes the Atomic Number is omitted from the nuclear notation since we already know Carbon has six protons from the atomic number on the periodic table. The oxidation number of an atom is indicated by a superscript in roman numerals on the right side of the chemical symbol. 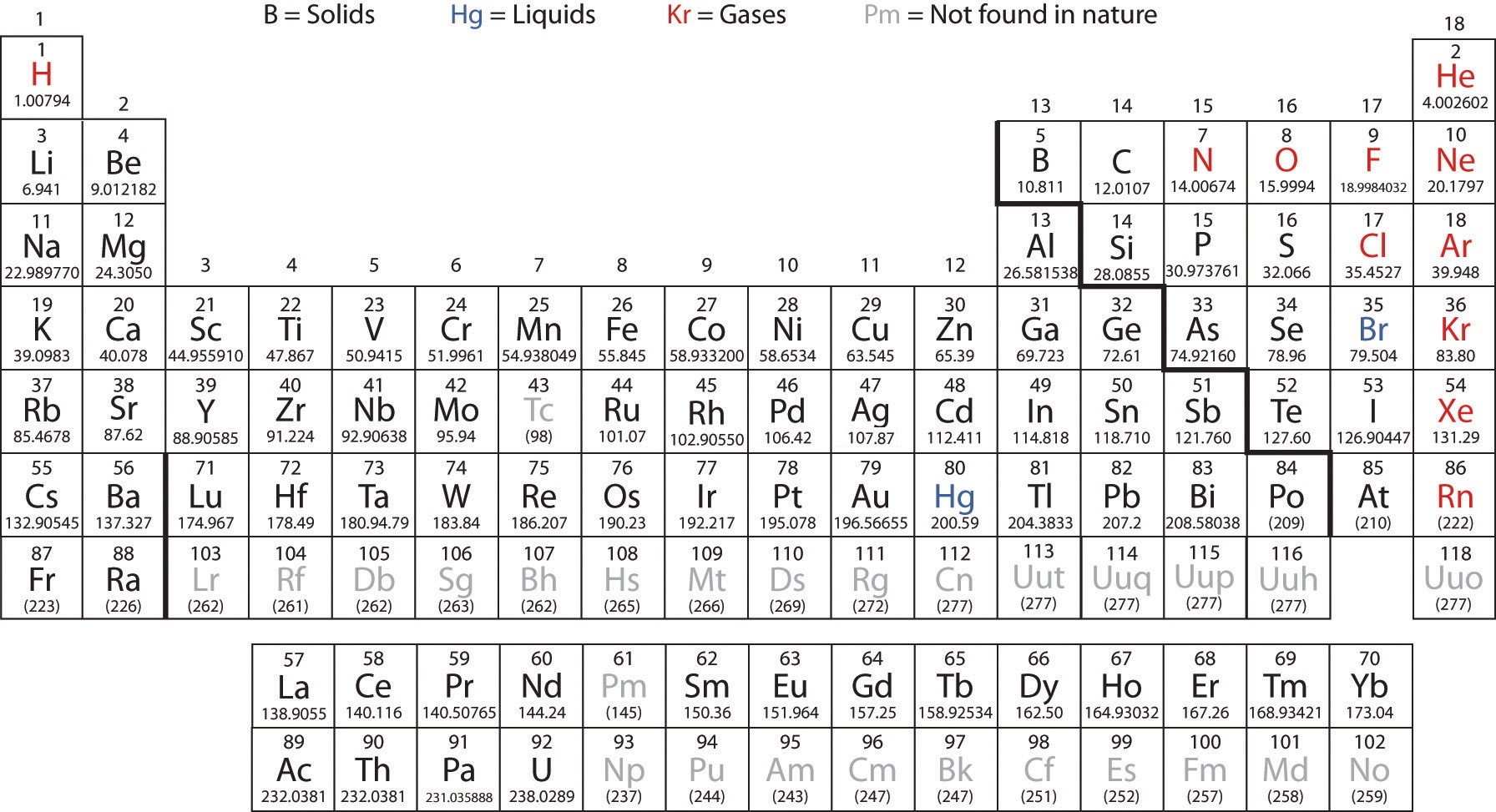
* Coming to the given element in the question. The highest superscript tells you the maximum number of electrons that are found in a particularatomic orbital.Do thep,d, andfblocks follow the same pattern. For the Periodic Table, the Atomic Number is on top and the average atomic mass is on the bottom. Hint: The atomic number of an element should be written in the subscript and mass number (number of protons number of neutrons) should be written in the superscript of the element when we are going to represent the symbol of an element. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
